INVESTIGATION OF FLUORIDE PRECIPITATION MECHANISMS AND OPTIMIZATION STRATEGIES FOR MINE DRAINAGE REMEDIATION
DOI:
https://doi.org/10.71146/kjmr890Keywords:
Fluoride removal, mine drainage, chemical precipitation, PHREEQC, optimization, RSM, calcium chloride, lanthanum chloride, water treatmentAbstract
Background:
Fluoride pollution in the mine drainage is of the most serious concern to water chemistry, human health, and aquatic ecosystem. Being present in much higher concentrations than World Health Organization (WHO) guidelines, require efficient and easy-to-implement remediation techniques, particularly in areas affected by phosphate, coal and uranium mines.
Aim: In this study, chemical and statistical methods are used to examine the mechanism of fluoride precipitation and present an optimally designed treatment process for mine drainage treatment.
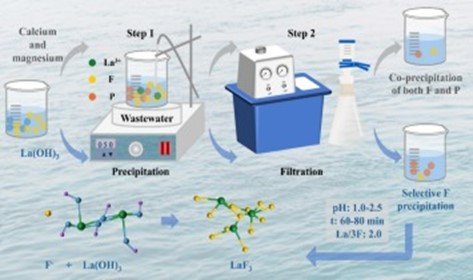
Method: Some of the precipitation agents such as calcium, aluminum, and lanthanum were studied at lab scale on batch mode at different pH, dosages, and contact time. the pre- and post-treatment fluoride levels were determined by ion-selective electrodes. From them it was possible to interpret the saturation and precipitation of fluoride minerals as reportedoftheseauthors. thermodynamic computations and kinetic analysis using PHREEQC were runed. Treatment parameter optimization was performed using Response Surface Methodology (RSM), and field verification was carried out for mine water samples from three locations.
Results: The optimal conditions of calcium and lanthanum salts removal of fluoride were obtained, and the fluoride removal rate of lanthanum chloride could be up to more than 99%. The best results of precipitation were found at pH 6.5–7.5 and prolonged contact time yielded little increases after 60 min. The experimental results were also in agreement with the thermodynamic, statistical models and then validated in the field (~93% fluoride rejection efficiency to various mine drainage samples).
Conclusion: The application of fluoride precipitation for mine water treatment is considered to be a suitable, cost-effective, and flexible process, particularly if it is optimized by statistical and geochemical modeling tools as were used in the present study.
Downloads
References
Alemayehu, M. M., Gebrehiwot, B., & Negash, M. (2023). Fluoride in groundwater and the potential health risk: A review. Environmental Advances, 13, 100266. https://doi.org/10.1016/j.envadv.2023.100266
Al-Maadheed, S., Amini, A., & Sharma, V. K. (2021). Fluoride removal from water by various techniques: A review. Chemosphere, 263, 128314. https://doi.org/10.1016/j.chemosphere.2020.128314
APHA. (2022). Standard Methods for the Examination of Water and Wastewater (24th ed.). American Public Health Association.
Ayanda, O. S., Fatoba, O. O., & Adekola, F. A. (2023). Optimization of fluoride removal from aqueous solution using natural adsorbents: A response surface methodology approach. Scientific Reports, 13, 11470. https://doi.org/10.1038/s41598-023-38607-w
Bhatnagar, A., & Kumar, E. (2023). Insights into the behavior of fluoride precipitation using calcium and aluminum salts in the presence of interfering ions. Journal of Water Process Engineering, 51, 103360. https://doi.org/10.1016/j.jwpe.2022.103360
Chouhan, S., & Flora, S. J. S. (2022). Arsenic and fluoride contamination in groundwater: A global overview with emphasis on remediation strategies. Environmental Toxicology and Pharmacology, 92, 103867. https://doi.org/10.1016/j.etap.2022.103867
He, Y., Liu, C., & Yu, J. (2022). Calcium-based precipitation for fluoride removal: Efficiency, mechanisms, and optimization. Journal of Environmental Chemical Engineering, 10(3), 107268. https://doi.org/10.1016/j.jece.2022.107268
Kumar, S., Tiwari, S., & Rai, P. K. (2022). Field-scale removal of fluoride from mine water using low-cost treatment techniques. Environmental Nanotechnology, Monitoring & Management, 18, 100729. https://doi.org/10.1016/j.enmm.2022.100729
Li, X., Zhang, Z., & Wu, Z. (2024). Optimizing fluoride removal using response surface methodology in the presence of multi-ion systems. Environmental Research, 234, 117225. https://doi.org/10.1016/j.envres.2024.117225
Meng, L., Zhao, C., & Fang, Y. (2023). A hybrid precipitation–adsorption method for fluoride removal from mining effluents. Water Research, 238, 120274. https://doi.org/10.1016/j.watres.2023.120274
Singh, R., Patel, A., & Sharma, A. (2024). Comparative assessment of fluoride removal using calcium and lanthanum salts in acid my drainage. Ecotoxicology and Environmental Safety, 266, 115354. https://doi.org/10.1016/j.ecoenv.2023.115354
Sun, Q., Yang, Z., & Wang, X. (2022). Application of rare earth elements for fluoride precipitation in industrial wastewater. Separation and Purification Technology, 282, 120030. https://doi.org/10.1016/j.seppur.2021.120030
Tang, Y., Liu, W., & Jiang, L. (2021). Modeling and optimization of fluoride removal from water using CaCl₂ via RSM. Environmental Technology & Innovation, 22, 101469. https://doi.org/10.1016/j.eti.2021.101469
Wang, L., Zhao, Y., & Li, J. (2024). Geochemical analysis and remediation strategies for fluoride-contaminated mine water in northern China. Journal of Hazardous Materials, 456, 131784. https://doi.org/10.1016/j.jhazmat.2023.131784
WHO. (2023). Guidelines for drinking-water quality (5th ed.). World Health Organization.
Zhao, Z., Lin, Y., & Hu, H. (2021). Fluoride removal in high-sulfate mine drainage using calcium-based treatment: Challenges and solutions. Journal of Environmental Management, 297, 113353. https://doi.org/10.1016/j.jenvman.2021.113353
Downloads
Published
License
Copyright (c) 2026 Aqsa Ashiq, Rohul Amin, Engr Dr Sana Saeed, A D' Yahcole M Morris (Author)

This work is licensed under a Creative Commons Attribution 4.0 International License.






